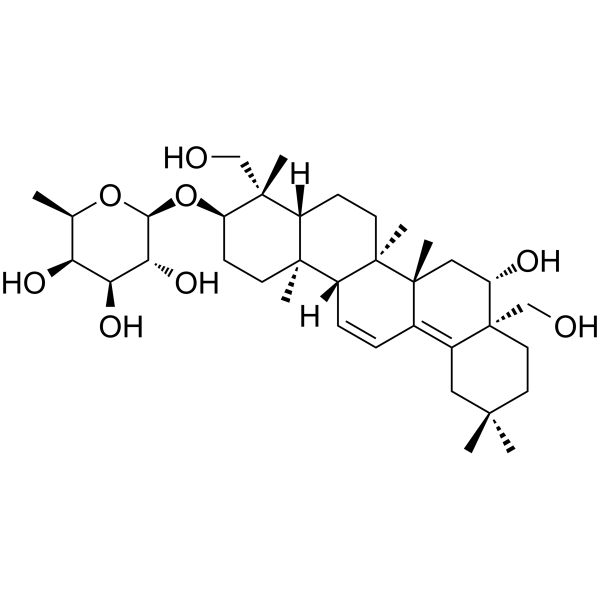
Prosaikogenin D
CAS No. 103629-72-7
Prosaikogenin D( Prosapogenin D )
Catalog No. M29246 CAS No. 103629-72-7
Prosaikogenin D is a natural product extracted from the roots of Buleurum bicaule Helm (Apiaceae) with anti-cancer activity.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 293 | Get Quote |


|
| 10MG | 437 | Get Quote |


|
| 100MG | Get Quote | Get Quote |


|
| 200MG | Get Quote | Get Quote |


|
| 500MG | Get Quote | Get Quote |


|
| 1G | Get Quote | Get Quote |


|
Biological Information
-
Product NameProsaikogenin D
-
NoteResearch use only, not for human use.
-
Brief DescriptionProsaikogenin D is a natural product extracted from the roots of Buleurum bicaule Helm (Apiaceae) with anti-cancer activity.
-
DescriptionProsaikogenin D is a natural product extracted from the roots of Buleurum bicaule Helm (Apiaceae) with anti-cancer activity.(In Vitro):Prosaikogenin D (20 - 60 μg/mL) suppresses the proliferation of A549 cells (IC50 = 57.2 μg/mL).
-
In VitroProsaikogenin D (20, 40, 60 μg/mL) suppresses the proliferation of A549 cells with an IC50 of 57.2 μg/mL.
-
In Vivo——
-
SynonymsProsapogenin D
-
PathwayOthers
-
TargetOther Targets
-
Recptor——
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number103629-72-7
-
Formula Weight618.852
-
Molecular FormulaC36H58O8
-
Purity>98% (HPLC)
-
Solubility——
-
SMILES[H][C@@]12CC[C@]3(C)[C@]([H])(C=CC4=C5CC(C)(C)CC[C@]5(CO)[C@@H](O)C[C@@]34C)[C@@]1(C)CC[C@@H](O[C@@H]1O[C@H](C)[C@H](O)[C@H](O)[C@H]1O)[C@]2(C)CO
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference



-
Emodin-1-O-β-D-gluco...
Emodin 1-O-beta-D-glucoside has neuroprotective and uncoupling activities, and that it may be the a new uncoupler of nNOS-PSD-95.
-
Cyclo(Tyr-Pro)
Cyclo(Tyr-Pro) shows antibacterial activity towards several marine bacterial species, it also shows weak antagonistic activity against VEGFR2 -CD. Cyclo(Tyr-Pro) and cyclo(Pro-Val) are toxic to both suspension cells and seedlings of Pinus thunbergii, which may offer some clues to research the mechanism of pine wilt disease caused by pine wood nematode.
-
9-(2,2-Dicyanovinyl)...
9-(2,2-Dicyanovinyl)julolidine (DCVJ) is a fluorogenic dye that is considered a fluorescent molecular rotor because its properties depend on the rotational relaxation of the molecule, which can be influenced by the viscosity of the solvent used.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


